AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Charge of electron in ergs1/2/2024  
This page is shared under a CK-12 license and was authored, remixed, and/or curated by Melissa Alviar-Agnew, Henry Agnew, and Lance S. Potential, statvolt, 299.8 Magnetic Field, Gauss, 10 Further units: 1 eV 1.602.10-12 erg 1 Ry 13.6 eV (ionization energy of. The atomic mass unit (amu) is a unit of mass equal to one-twelfth the mass of a carbon-12 atom.As a result, a neutral atom must have an equal number of protons and electrons. The positive charge on a proton is equal in magnitude to the negative charge on an electron.Protons and neutrons have approximately the same mass, but they are both much more massive than electrons (approximately 2,000 times as massive as an electron). 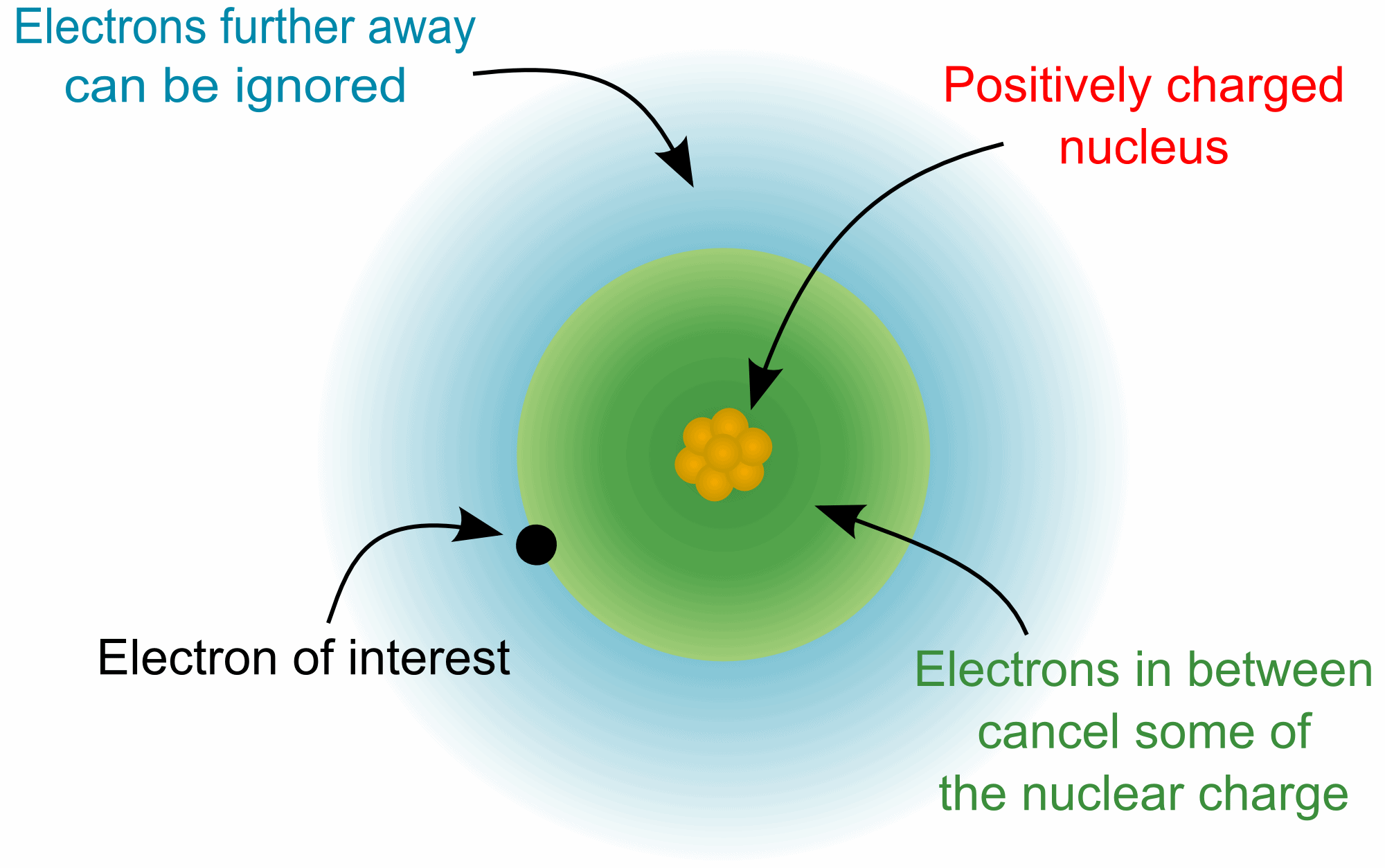
Like protons, neutrons are bound into the atom's nucleus as a result of the strong nuclear force. Neutrons are a type of subatomic particle with no charge (they are neutral).Protons are bound together in an atom's nucleus as a result of the strong nuclear force. Protons are a type of subatomic particle with a positive charge.Electrons are a type of subatomic particle with a negative charge.The energy transferred by a wave in a given time is called its intensity.\): Properties of Subatomic Particles Particle īefore Einstein's paper, electromagnetic radiation such as visible light was considered to behave as a wave: hence the use of the terms "frequency" and "wavelength" to characterize different types of radiation. The Nobel committee awarded the prize for his work on the photo-electric effect, rather than relativity, both because of a bias against purely theoretical physics not grounded in discovery or experiment, and dissent amongst its members as to the actual proof that relativity was real. Einstein's 1905 paper discussing the effect in terms of light quanta would earn him the Nobel Prize in 1921, after his predictions had been confirmed by the experimental work of Robert Andrews Millikan. In relation to the base unit of electric charge > (coulombs), 1 Electron Charge (e-) is equal to 1.6021766208E-19 coulombs, while 1 Esu Of Charge (esu) 3.335640951982E-10 coulombs. Another particularly thorough investigation was published by Philipp Lenard (Lénárd Fülöp) in 1902. It was first observed by Alexandre Edmond Becquerel in 1839, although credit is usually reserved for Heinrich Hertz, who published the first thorough investigation in 1887. The photoelectric effect is the emission of electrons (called "photoelectrons") from a surface when light is shone on it. The first Solvay Conference in 1911 was devoted to "the theory of radiation and quanta". They contributed greatly (along with Einstein's work on the photoelectric effect) in convincing physicists that Planck's postulate of quantized energy levels was more than a mere mathematical formalism. 2 This elementary charge is a fundamental physical constant. Definition: In relation to the base unit of electric charge > (. These proofs are commonly known as the " ultraviolet catastrophe", a name coined by Paul Ehrenfest in 1911. The elementary charge, usually denoted by e, is the electric charge carried by a single proton or, equivalently, the magnitude of the negative electric charge carried by a single electron, which has charge 1 e. Always check the results rounding errors may occur. The black-body problem was revisited in 1905, when Lord Rayleigh and James Jeans (on the one hand) and Albert Einstein (on the other hand) independently proved that classical electromagnetism could never account for the observed spectrum. The observed Planck curves at different temperatures, and the divergence of the theoretical Rayleigh–Jeans (black) curve from the observed Planck curve at 5000K. M L 2 T − 1 from the same data and theory. H h, or ℏ \hbar for the reduced Planck constant electron volt, unit of energy commonly used in atomic and nuclear physics, equal to the energy gained by an electron (a charged particle carrying unit. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
